

Whereas metals in group I and II of the periodic table are only oxidized by one and two electrons, respectively, transition (d-block) metals are capable of achieving multiple oxidation states. transition metals new classes of compounds were made, their chemical behaviour studied and the bonding in the com- pounds investigated.
#Transition metals reactivity series
These observations, as well as thermodynamic investigations, suggest that late metal-heteroatom bonds are strongly polarized and possess significant ionic character, properties that play an important role in their interactions with organic compounds. When discussing the chemistry of transition metal complexes, it is customary to determine the oxidation state of the metal and the number of valence shell. In Unit IV, we continue to consider chemical reactions but now focus on reactions that involve the loss (oxidation) and gain (reduction) of electrons. In particular, the first transition series metal complexes are expected to react more rapidly than heavier element analogs and possess special freedom to. Indeed, CO2 activated on 3-S of Fe6Mo2S8 (SEt)3 (5-) is fixed at the carbonyl carbon of thioesters trapped on a neighboring iron of the cluster, and -keto acids are produced catalytically.

Warning: Can only detect less than 5000 charactersÑ ñ,¸¸. Structurally robust 3-S of reduced metal-sulfur clusters provides a suitable site for reductive activation of CO2 with retention of the framework. The M-X complexes typically exhibit nucleophilic reactivity, in some cases form strong hydrogen bonds to proton donors, and even deprotonate relatively weak acids. Describe the reactivity series of transition metals. The heteroatoms in these materials form surprisingly strong bonds to their metal centers, and their bond energies do not necessarily correlate with the energies of the corresponding H-X bonds. Their reactivity, dependent on many factors such as. As with all metals, the transition elements are both ductile and. New synthetic strategies have now made possible the isolation and study of a variety of monomeric late-metal alkoxide, aryloxide, and amide complexes, including parent hydroxide and amide species. Moreover, laboratory studies have shown that transition metal ions (TMI) are highly reactive in solution. The 38 elements in groups 3 through 12 of the periodic table are called transition metals. Fe(H2O)6 3+ + 3 acac- Fe(acac) 3 + 6 H2O Cu(H2O)6SO4 + 4 NH3 Cu(NH3)4(H2O)2SO4 2) Addition (dissociation) reactions: MLn + L’ MLnL’ egs. Furthermore, high degrees of diastereo- and enantioselectivity have been observed in several cases. Frustration in these structures arises from the fixed location of both the Lewis acid and the base on the surfaces. Recent reports have shown that CH, NH, BH, OH, SH, SiH, GeH, SnH and PH insertion reactions are feasible with a variety of transition metal catalysts, both inter- and intramolecularly. This behavior can be explained due to the intrinsic acidic (metallic ion sites) and basic atoms (oxide units) present within the surface (Scheme 92 ).
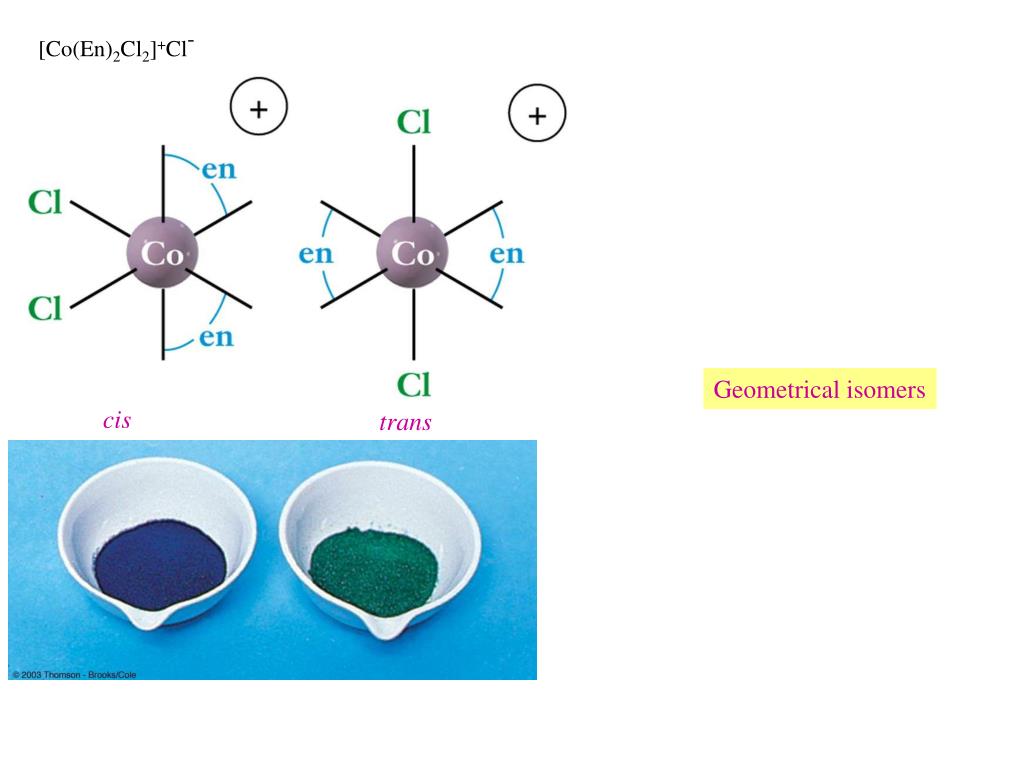
26) Four main types of reactivity: 1) Substitution reactions: MLn + L’ MLn-1L’ + L egs. Transition metal oxide nanostructures can exhibit FLP-like reactivity. The combination of the redox-active ligand scaffold, first-row transition metal center, and intramolecular, multi-center hydrogen bonding interaction made the. Keywords: amorphous silica density functional calculations reactivity indices single-atom catalysis transition metals. Despite this, our understanding of such M-X linkages still lags behind that of their M-H and M-C analogues. Reactivity of Transition Metal Complexes (H&S 3rd Ed., Chpt. Finally, the obtained Fermi weighted density of states reactivity trends show a good agreement with the chemical characteristics of the investigated metal atoms as well as the experimental data. \( \newcommand\).Complexes containing bonds between heteroatoms such as nitrogen and oxygen and "late" transition metals (i.e., those located on the right side of the transition series) have been implicated as reactive intermediates in numerous important catalytic systems.

